Overview technologies

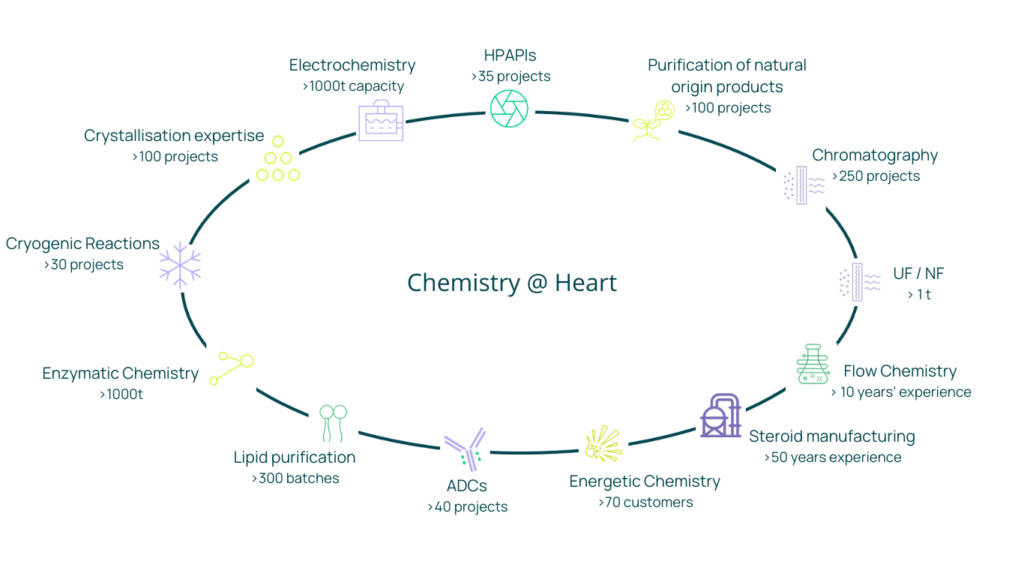
The increasing complexity of today’s clinical pipeline often requires expertise in specialized and innovative technologies. Customers are embracing alternative, state-of-the-art solutions in which reliability, sustainability & value creation are paramount.
Novasep CDMO is a world leader in several technologies including industrial chromatography, hazardous chemistry, antibody drug conjugates, and electrochemistry, supported by an extensive toolbox of classical chemistry.
The key to success is our ability to seamlessly scale up these technologies under full cGMP at a wide range of scales, leveraging our network of in-house scientific expertise at our centers of excellence, with specialized development and manufacturing facilities.
Deep expertise & capabilities